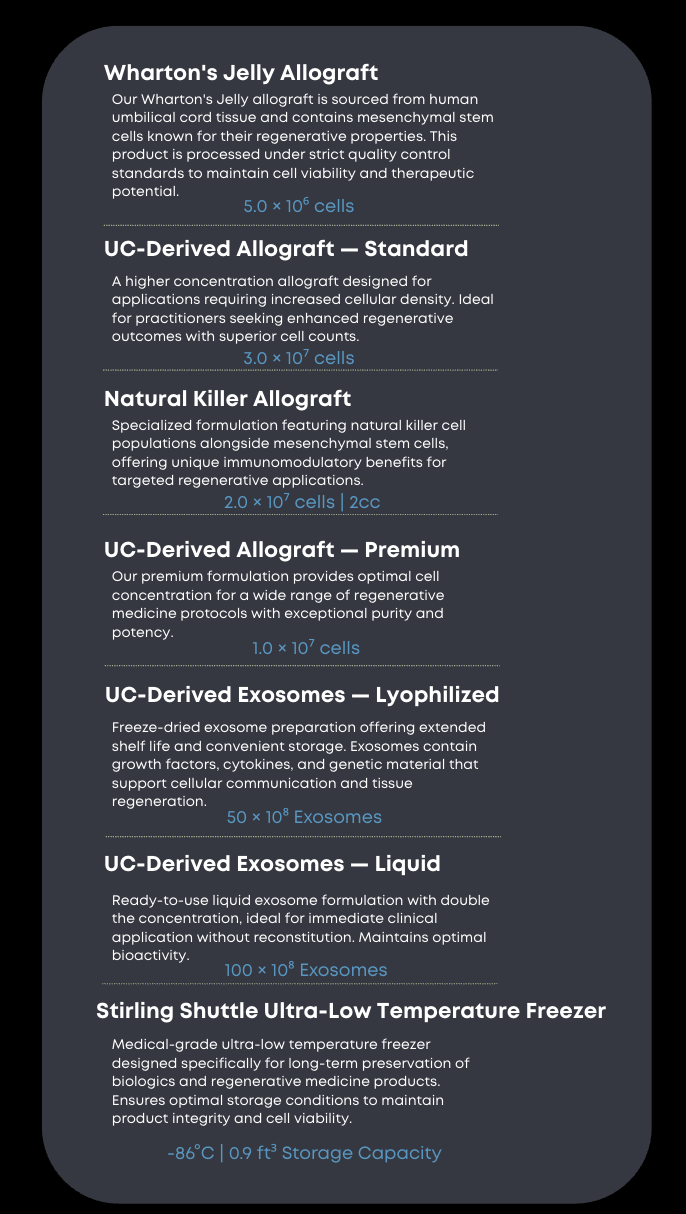
Products

Quality Assurance & Compliance
Our laboratory adheres to stringent guidelines in accordance with Current Good Manufacturing Practices (cGMP), Current Good Tissue Practice (cGTP), State Regulations, AATB guidelines, and FDA requirements for Human Cells, Tissues, and Cellular and Tissue-based Products (HCT/P).
Every product undergoes rigorous testing and quality control to ensure safety, purity, and potency. We are committed to maintaining the highest standards in regenerative medicine manufacturing.